Think of an atom as a ______________________________
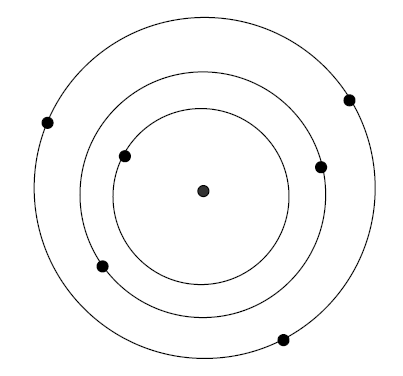
__________________________________________
1. __________ _____________ an electron in an atom are
_________
2. Each ______________is a ______________ __________________
3.
4. When ____________ in an atom ________________________
• ____________________________________
• ____________________________________
Permitted Orbital Energies for Hydrogen - Reference table
Each orbital is associated with a specific energy
|
Energy Level # |
Energy of this level |
|
|
n=3 |
_________ |
_________State
|
|
n=2 |
_________ |
_________State
|
|
n=1 |
_________
|
_________State _________ Energy Level |
- Atoms can only absorb or emit energy
that is equal _______________
____________________________________________________________
- Electrons can be
____________________________________________________
__________________________________________________________________
-
_______________________________________________________________
-
_____________________________________________
-
_______________________________________________________________
| Example - How much
energy is
absorbed when an electron in hydrogen moves from the
energy
levels n = 2 to n = 5?
How much energy is needed for an electron in the n=1 energy level to move to the n = 2 energy level? n =1 ___________
Convert ________________ page 1 Reference 1 eV = _______________________ E = ______________________ = ______________________ = ______________________
5) Ionization Energy -
n = 2 n = 3 Why were electrons limited in the energies they can possess?
Possible electron circumference = _____________________________ where n=1,2,3,4 etc The ____________________________ of an electron is ________ And λ = ____________________________
|