![]()
 Home
Home
![]()

Elastic PE
|
If the force you apply to a spring is directly proportional to stretch, how many times would you have to increase your force to get 4 times the stretch? |
Show Me The Physics
YouTube
Channel
Elastic P.E. - work stored in a deformed spring
a) General Equation
PEs = Fx
F = Average Force needed to deform spring
x = distance deformed (stretch or compress)

Ideal Spring
showmethephysics.com
b) Hooke's Law - force needed to deform an ideal spring, a given amount is directly proportional to its displacement (deformation)

Graphic from Learning-Connections Clipart
![]()
Robert Hooke
'Newton's bitter rival'
![]()
Ex) Ideal Spring
|
F
(Force to Deform) (N) |
X
(distance deformed) (m) |
|
2.0 |
.30 |
| 4.0 | .60 |
|
6.0 |
.90 |
|
8.0 |
1.2 |
![]()
University of Colorado - PhET
![]()
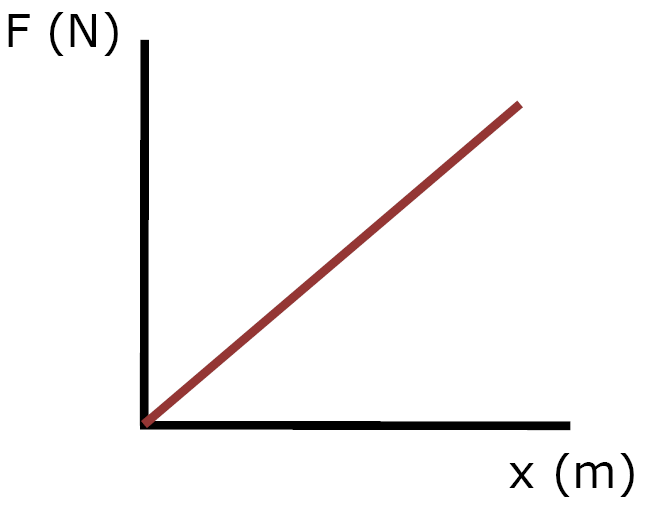
slope = F/x
x = stretch
c) Spring Constant (k), Ideal Spring
- quantifies stiffness of a spring
| K | = | F | (N) |
| X | (m) |
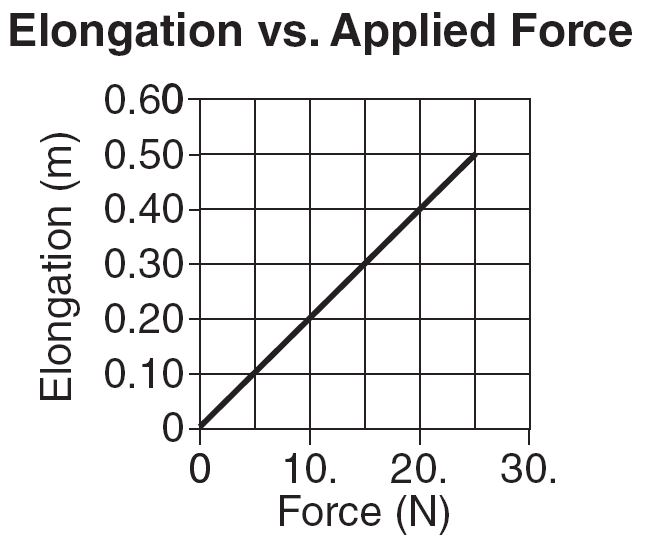
What is the spring constant of the spring that produced the data? above?
| F
(Force to Deform) (N) |
X
(distance deformed) (m) |
| 2.0 | .30 |
| 4.0 | .60 |
| 6.0 | .90 |
| 8.0 | 1.2 |
| k | = | 2.0 N |
| .30 m |
k = 6.7 N/m
Which is the stiffer spring?
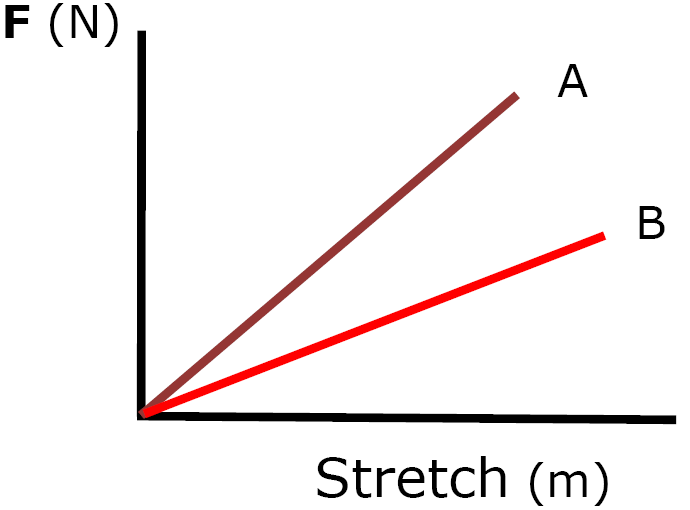
Spring A is the stiffer spring.
More
force is required
for the same stretch

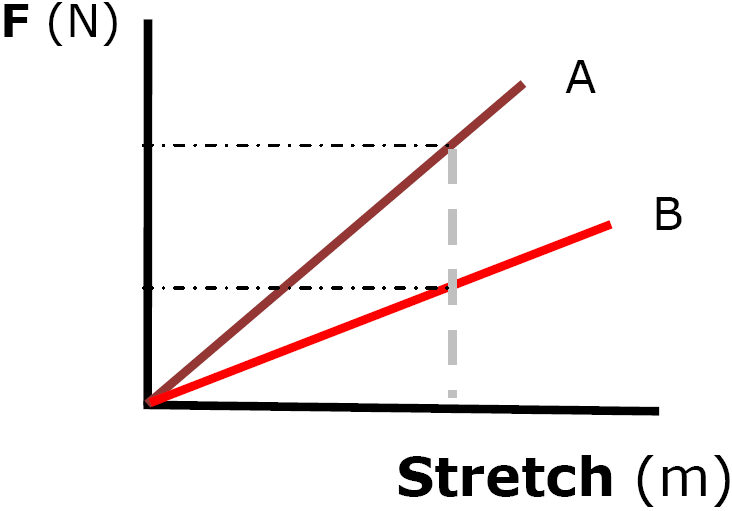
Ex) Plots and K
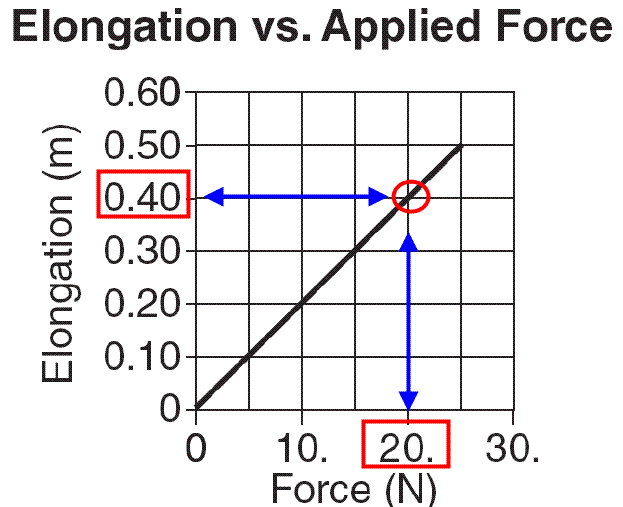
Pick any point on the line
| K | = | F | (N) |
| X | (m) |
| K | = | 20. N |
| .40 m |
K = 50. N/m
d) Elastic P.E. = area of triangle under F vs x graph
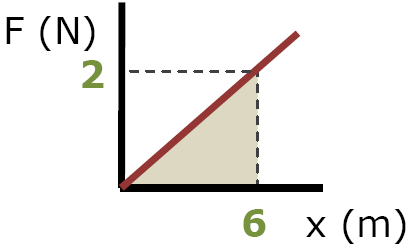
PEs = (½)basexheight
PEs = 6 Joules
![]()
![]()
Try this at home!
What crushed this can?
S.M.U. Physics
The can implodes because the atmospheric pressure is greater than the pressure inside the can.
When the can was submerged in the water it caused the water vapor inside the can to rapidly cool. When a gas is cooled, its pressure goes down. This drop in pressure causes the pressure inside the can to be lower than the atmospheric pressure.
Note: Use less than 1 cm of water in this experiment (about 1/2 inch)
©Tony Mangiacapre.,
- All Rights Reserved [Home]
Established 1995
Use any
material on this site (w/ attribution)