|

 Home
Home


Bohr's
Conclusions
|
When an atom GAINS energy,
it is said to be an _______ state
|

The Aurora Borealis, or Northern Lights,
shines above Bear Lake, Eielson Air Force Base, Alaska
gallery
(Full
Image)
United
States Air Force photo by Senior Airman Joshua Strang

Fill In The Blank Notes
Bohr's Conclusions
Think of an atom as a
miniature solar system
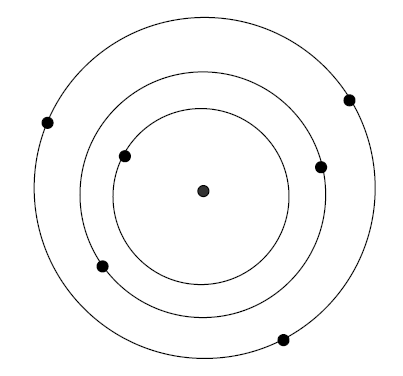
3 orbits are permitted
for this atom
1. Permitted orbits
of an electron in an atom are
limited
2. Each orbit
is a
different energy
3. Inside orbits
have
lower energies than the outside ones
4. When electrons
in an atom
change energy states they
absorb or release energy
 Up energy
level,
Up energy
level,
energy absorbed
 Down
energy level, Down
energy level, 
energy released
as light
Permitted Orbital Energies
for Hydrogen
Reference table

Each orbital is associated with a specific energy
|
Energy
Level # |
Energy |
|
|
n = 3 |
-1.51 eV |
Excited State
|
|
n =
2 |
-3.40 eV |
Excited State
|
|
n =
1 |
-13.6 eV |
Ground
State Lowest Energy Level |
Electrons can only
absorb or emit energy that is
equal to the difference between the absolute value of
2 energy levels.
A single photon of energy is
given off when electrons
go down energy levels.


Tony Mangiacapre.,- All Rights Reserved [Home]
Established 1995
Use any material on this site (w/ attribution)
|