![]()
![]()

Electron Energies?
|
When electrons return to the ground state they give off energy in the form of _______ |
![]()

Why were electrons limited
in the energies they can possess?
Electron Standing Waves
School blocks YouTube? Use the file below.
St. Mary's U. Astronomy and Physics Dept.
1. An electron is a standing wave. (A bound wave)

5 wavelengths
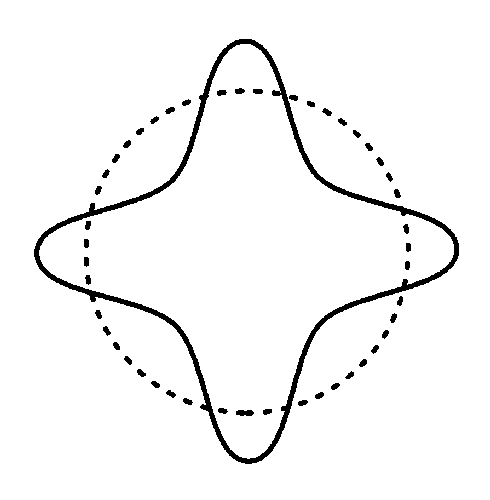
4 wavelengths
3 wavelengths
2 wavelengths
2. Circular standing waves can only be a whole number of wavelengths
![]()
Monterey Institute for Technology and Education
![]()
3. Limited wavelengths mean limited possible energies
Possible Electron Circumferences
= 2∏r = nλ
where n = 1,2,3,4 etc
Wavelength - describes probability of finding an electron at particular position.
Electron is a wave circled in on itself
This standing wave has four cycles

The angular (circular) momentum of
an electron is limited ...
...because a circular electron wave must be a whole number of wavelengths
Then C = 2(∏)r = nλ
where n = 1,2,3,4 etc
And λ = h/p = h/mv
Then … 2(∏)r = nh/mv
Angular Momentum
=
mvr = n[h/2∏]
| Angular momentum (mvr) is restricted to whole number multiples of h/2∏ |
![]()
![]()
©Tony Mangiacapre., - All Rights Reserved
[Home]
Established 1995
Use any
material on this site (w/ attribution)